The answer to this question is ABSOLUTELY. Yes. 100%.
However, the amount and type of carbohydrates someone can eat and maintain regulated blood sugar is highly individual [1].
To really dig into this question we must first define what it means to be objectively metabolically healthy in terms of blood glucose control specifically:
- A fasting glucose of 75 to 90ish mg/dL with a mean amplitude glucose excursion (MAGE) of 40 to 50 mg/dL are probably fairly solid lines in the sand [2, 3].
- One also likely wants to maintain a Hemoglobin A1c (HbA1c) of 5.5 or less [1]. Hemoglobin A1c is a more long-term marker of blood glucose control and is essentially a measure of the damage that high blood glucose is doing to hemoglobin within your red blood cells, which generally live for about 90 to 120 days (if red blood cells live longer or shorter that can influence the interpretation of an HbA1c value).
These numbers and acronyms may look like hieroglyphics to you, so it is likely best to begin this discussion by contrasting two extreme scenarios.
To do this, let’s think about the blood glucose control of someone living with type 2 diabetes who is by definition metabolically unhealthy and metabolically inflexible.
VS.
An extremely metabolically flexible athlete with a lot of muscle mass at the low end of the body fat spectrum.
The diagnostic criteria for type 2 diabetes is a fasting blood sugar ≥ 126 mg/dL, a HbA1c ≥ 6.5, and/or a blood glucose value ≥200 mg/dL two hours after an oral glucose tolerance test (OGTT).
Below one can see some scenarios of what this might look like with OGTT data. The renal threshold is when we start blowing glucose out of our kidneys.
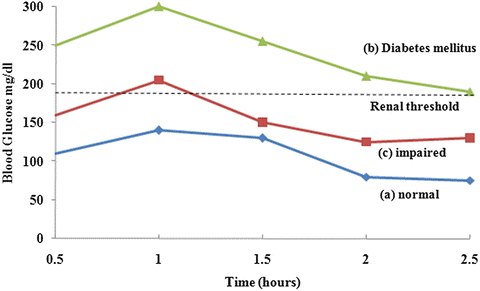
As another example that isn't depicted in the chart above, if a person is just sneaking into the type 2 diabetes criterion and starts the test at 130 mg/dl that equals about 6.5 grams in the entire bloodstream, given that most humans have about 5 liters of blood.
Then this individual drinks 75 grams of glucose in the form of a pink chalky liquid and two hours later they are right at 200 mg/dl which equals about 10 grams in the entire bloodstream.
10 - 6.5 = only 3.5 grams of extra blood glucose?!?
Yup…if we do the maths you’ll notice that even someone with type 2 diabetes can still dissipate 71.5 grams of the 75 grams in two hours.
Well, not exactly because only about 75% of that 75 grams will hit systemic circulation within 3.5 hours [5], but even in this type 2 diabetes scenario this individual was still be able to dissipate at least ~40-50 grams of glucose from the OGTT.
This example shows us how tightly human beings regulate blood glucose dynamics even in overt disease classifications.
At this point, someone might be thinking…well...all that doesn’t sound so bad.
The problem is that above 160-180 mg/dl we are getting into what is objectively known as hyperglycemia [6]. Despite what the internet may say sugar as a compound is innocuous, not bad, no problem [7, 8], but hyperglycemia is a well-established and well-known dumpster fire of negative physiologic effects including:
- Increased systemic inflammation.
- Endothelial dysfunction and increased risk of cardiovascular disease.
- The glycation and damage to our neurons which can lead to neuropathies and increased risk of dementia.
- Frying our retinas, which can lead to blindness.
- Blasting our kidneys with high glucose which can lead to chronic kidney disease and potentially dialysis.
- Diabetic foot ulcers.
- Diabetic ketoacidosis, which can potentially lead to coma and even death.
If we are throwing out the word health, we likely do not want to live our lives in a hyperglycemic state.
Now let’s contrast the type 2 diabetes scenario above with an athlete who never leaves the range of 80 to 120 mg/dl, while consuming roughly 460 grams of carbohydrates!
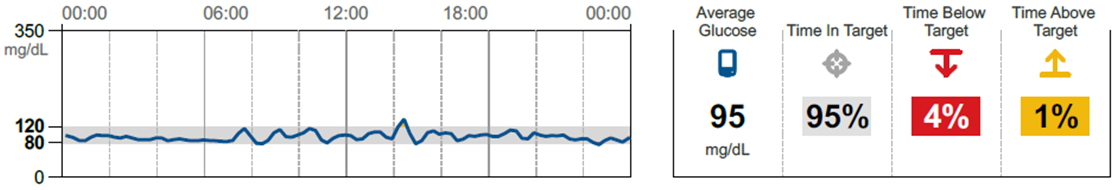
What this chart shows is that even in the face of bountiful amounts of rice and even rice cakes throughout the day, this individual’s body was able to maintain blood glucose within a two-gram window!
This can sound utterly ridiculous to be able to eat this amount of carbohydrates and maintain such tight blood glucose control, but we have seen this time and time again in healthy, well-muscled training individuals.
This is anti-fragility and resilience in the face of our current food environment.
This is metabolic flexibility, and IMO the pinnacle of metabolic health.
This generally requires large amounts of muscle, consistent training, movement outside the gym, and staying well within one's personal fat threshold.
If we look at these examples and the literature, the general heuristics that can help one maintain better blood sugar control and metabolic flexibility are:
Number 1 - getting into and maintaining the metabolically healthy body composition numbers that we have discussed HERE will likely help maintain better baseline blood glucose control.
Now, if someone is metabolically inflexible or metabolically unhealthy they are likely not going to process large amounts of carbohydrates very well and what can be helpful in this scenario is:
- Limiting the total amount of carbohydrates at the meal to 30-50 grams [9].
- Eating whole food-based meals that contain both fat and protein [10, 11].
- Exercise…specifically HIIT or Resistance Based Higher Rep Circuit Training [12-15]
- Moving consistently throughout the day [16, 17].
Just to show you one of the wildest effect sizes I have ever seen when it comes to manipulating blood glucose levels check out the circuit training continuous glucose monitoring study by Cruz et al., 2019 below [13]. This was a cross-over study design meaning that every person did each intervention.
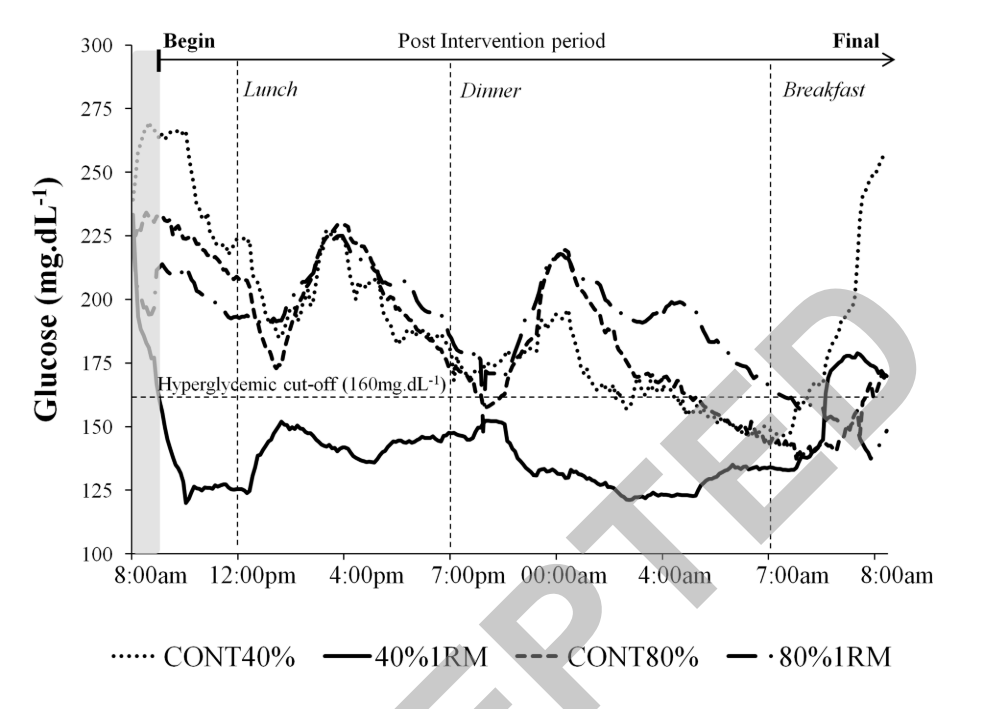
When individuals living with type 2 diabetes underwent high-repetition lower-weight (40% 1RM) circuit training, they pretty much had normal range glucose values for ~24 hours after training (bold line at the bottom of the chart above). This means they were transiently metabolically healthy, likely due to pulling energy through the system at the cellular level.
We can then see the line trend start to shoot back up again the next day which is in line with recent literature on the transitory nature of exercise’s effect on blood glucose control without a sustained energy deficit and subsequent body composition changes (fat loss and/or muscle gain) [18-20].
To summarize, the more metabolically healthy you are, the more leeway you likely have when it comes to carbohydrate consumption while staying within a 40-50 mg/dl window of blood glucose control (MAGE) and not going hyperglycemic. That said, even if you are metabolically healthy, eating a primarily whole food and nutrient-dense diet, and maintaining high levels of physical activity are some of the biggest keys to staying healthy throughout the life course.
It is also important to note that chronic disease is about dose and duration AKA high blood glucose, high blood lipids, high blood pressure, high inflammation, and/or genetic risk over time.
Thus, if you pop into the hyperglycemic range every once in a while it isn’t necessarily a reason to freak out and buy more life insurance. Also, if you are trying to make gains as an extremely advanced trainee, a touch of pre-diabetes might be worth it to you. This is an individual risk/reward decision.
If you are curious about what your daily blood glucose control might look like you can work with a medical provider to insert a continuous glucose monitor to check your blood glucose response to your normal meals in your normal daily life.
Continuous glucose monitoring can provide maybe might be able to provide some broad insights in some populations [21-24], but they can also lead to some very weird biohacker nonsense where people major in the minors and lose their marbles over perfectly normal blood glucose responses.
REFERENCES:
1. Zeevi, D., et al., Personalized Nutrition by Prediction of Glycemic Responses. Cell, 2015. 163(5): p. 1079-94.
2. Gude, F., et al., Glycemic Variability and Its Association With Demographics and Lifestyles in a General Adult Population. J Diabetes Sci Technol, 2017. 11(4): p. 780-790.
3. Kuroda, M., et al., Effect of daily glucose fluctuation on coronary plaque vulnerability in patients pre-treated with lipid-lowering therapy: a prospective observational study. JACC Cardiovasc Interv, 2015. 8(6): p. 800-811.
4. Kumar, V. and K.D. Gill, To Perform Glucose Tolerance Test, in Basic Concepts in Clinical Biochemistry: A Practical Guide, V. Kumar and K.D. Gill, Editors. 2018, Springer Singapore: Singapore. p. 63-66.
5. Ferrannini, E., et al., The disposal of an oral glucose load in healthy subjects. A quantitative study. Diabetes, 1985. 34(6): p. 580-8.
6. Blaak, E.E., et al., Impact of postprandial glycaemia on health and prevention of disease. Obes Rev, 2012. 13(10): p. 923-84.
7. Esposito, K., et al., Inflammatory cytokine concentrations are acutely increased by hyperglycemia in humans: role of oxidative stress.Circulation, 2002. 106(16): p. 2067-72.
8. Ceriello, A., et al., Glucose "peak" and glucose "spike": Impact on endothelial function and oxidative stress.Diabetes Res Clin Pract, 2008. 82(2): p. 262-7.
9. Blaychfeld-Magnazi, M., et al., The effect of a low-carbohydrate high-fat diet and ethnicity on daily glucose profile in type 2 diabetes determined by continuous glucose monitoring. Eur J Nutr, 2020. 59(5): p. 1929-1936.
10. Moghaddam, E., J.A. Vogt, and T.M. Wolever, The effects of fat and protein on glycemic responses in nondiabetic humans vary with waist circumference, fasting plasma insulin, and dietary fiber intake. J Nutr, 2006. 136(10): p. 2506-11.
11. Ballance, S., et al., Predicting mixed-meal measured glycaemic index in healthy subjects. Eur J Nutr, 2019. 58(7): p. 2657-2667.
12. Munan, M., et al., Acute and Chronic Effects of Exercise on Continuous Glucose Monitoring Outcomes in Type 2 Diabetes: A Meta-Analysis.Front Endocrinol (Lausanne), 2020. 11: p. 495.
13. Cruz, L.C.D., et al., Low-Intensity Resistance Exercise Reduces Hyperglycemia and Enhances Glucose Control Over a 24-Hour Period in Women With Type 2 Diabetes. J Strength Cond Res, 2019. 33(10): p. 2826-2835.
14. Terada, T., et al., Targeting specific interstitial glycemic parameters with high-intensity interval exercise and fasted-state exercise in type 2 diabetes. Metabolism, 2016. 65(5): p. 599-608.
15. Ferrannini, E., et al., Adipose tissue and skeletal muscle insulin-mediated glucose uptake in insulin resistance: role of blood flow and diabetes. Am J Clin Nutr, 2018. 108(4): p. 749-758.
16. Loh, R., et al., Effects of Interrupting Prolonged Sitting with Physical Activity Breaks on Blood Glucose, Insulin and Triacylglycerol Measures: A Systematic Review and Meta-analysis. Sports Med, 2020. 50(2): p. 295-330.
17. Dempsey, P.C., et al., Interrupting prolonged sitting in type 2 diabetes: nocturnal persistence of improved glycaemic control.Diabetologia, 2017. 60(3): p. 499-507.
18. Schleh, M.W., et al., Energy Deficit Required for Exercise-induced Improvements in Glycemia the Next Day. Med Sci Sports Exerc, 2020. 52(4): p. 976-982.
19. Fisher, G., et al., Acute Effects of Exercise Intensity on Insulin Sensitivity under Energy Balance. Med Sci Sports Exerc, 2019. 51(5): p. 988-994.
20. Ross, R., et al., Exercise-induced reduction in obesity and insulin resistance in women: a randomized controlled trial. Obes Res, 2004. 12(5): p. 789-98.
21. Bailey, K.J., J.P. Little, and M.E. Jung, Self-Monitoring Using Continuous Glucose Monitors with Real-Time Feedback Improves Exercise Adherence in Individuals with Impaired Blood Glucose: A Pilot Study. Diabetes Technol Ther, 2016. 18(3): p. 185-93.
22. Ehrhardt, N. and E. Al Zaghal, Continuous Glucose Monitoring As a Behavior Modification Tool. Clin Diabetes, 2020. 38(2): p. 126-131.
23. Ehrhardt, N. and E. Al Zaghal, Behavior Modification in Prediabetes and Diabetes: Potential Use of Real-Time Continuous Glucose Monitoring. J Diabetes Sci Technol, 2019. 13(2): p. 271-275.
24. Yost, O., et al., Continuous Glucose Monitoring With Low-Carbohydrate Diet Coaching in Adults With Prediabetes: Mixed Methods Pilot Study. JMIR Diabetes, 2020. 5(4): p. e21551.
25. Freckmann, G., et al., Measures of Accuracy for Continuous Glucose Monitoring and Blood Glucose Monitoring Devices. J Diabetes Sci Technol, 2019. 13(3): p. 575-583.
26. Kanamori, K., et al., Postprandial Glucose Surges after Extremely Low Carbohydrate Diet in Healthy Adults. Tohoku J Exp Med, 2017. 243(1): p. 35-39.




Related Content
Is Hemoglobin A1c Useful In Athletes and How Is It Affected By Chronic Exercise?
May 28, 2025
Revisiting The Effect of Carbohydrate Timing and Low Energy Availability on Performance.
Jan 23, 2025
How Accurate Are Cooked Foods When Logging?
Aug 03, 2023